
Dual Compartment vs. Single Compartment Steroid Injection for de Quervain Disease

Dual Compartment vs. Single Compartment Steroid Injection for de Quervain Disease
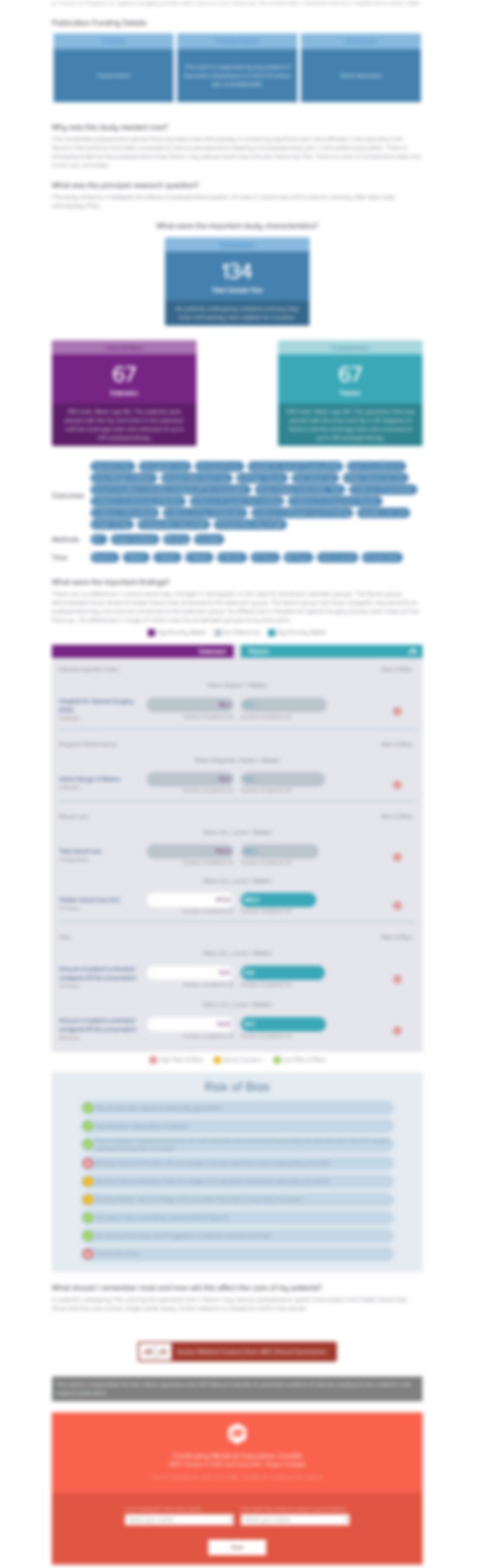
Is a Steroid Injection in Both Compartments More Effective than an Injection in the Extensor Pollicis Brevis Subcompartment Alone in Patients with de Quervain Disease? A Randomized, Controlled Trial.
Clin Orthop Relat Res. 2022 Apr 1;480(4): 762-770.Synopsis
50 patients with de Quervain disease were randomized to receive a steroid injection into the extensor pollicis brevis subcompartment only (n=25) or into both the abductor pollicis longus and extensor pollicis brevis subcompartments (n=25). The outcomes of interest included pain on a Visual Analog Scale (VAS) and the incidence of injection-related complications. Results revealed no significant diff...
To view the full content, login to your account,
or start your 30-day FREE Trial today.
FREE TRIAL
LOGIN
Forgot Password?
Explore some of our unlocked ACE Reports below!
Learn about our AI Driven
High Impact Search Feature
Our AI driven High Impact metric calculates the impact an article will have by considering both the publishing journal and the content of the article itself. Built using the latest advances in natural language processing, OE High Impact predicts an article’s future number of citations better than impact factor alone.
Continue



 LOGIN
LOGIN

Join the Conversation
Please Login or Join to leave comments.