
Rofecoxib and celecoxib demonstrate similar, significant 6-week efficacy vs placebo in OA treatment

Rofecoxib and celecoxib demonstrate similar, significant 6-week efficacy vs placebo in OA treatment
Efficacy and safety of rofecoxib 12.5 mg and celecoxib 200 mg in two similarly designed osteoarthritis studies
Curr Med Res Opin. 2006 Jan;22(1):199-210Synopsis
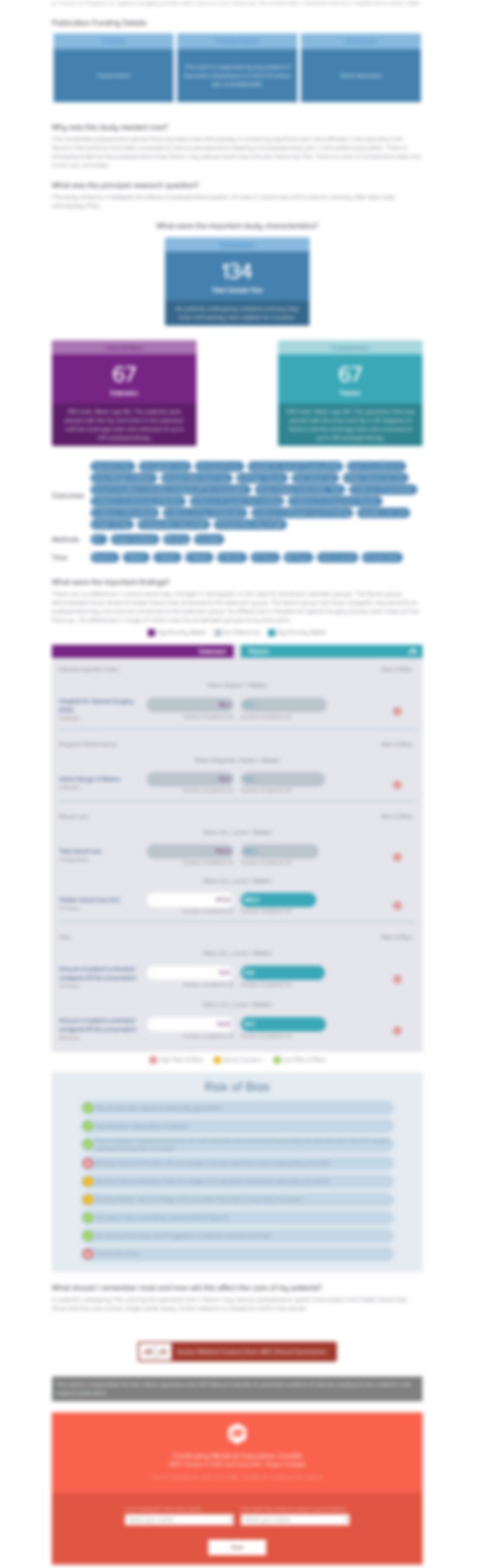
808 patients, combined from two trials, with knee osteoarthritis were randomized to receive either celecoxib 200mg daily, rofecoxib 12.5mg daily, or placebo. Patients were assessed over a 6-week period of treatment on a Patient Global Assessment of Response to Therapy (PGART), the Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) Pain and Physical Function subscales, and the i...
To view the full content, login to your account,
or start your 30-day FREE Trial today.
FREE TRIAL
LOGIN
Forgot Password?
Explore some of our unlocked ACE Reports below!
Learn about our AI Driven
High Impact Search Feature
Our AI driven High Impact metric calculates the impact an article will have by considering both the publishing journal and the content of the article itself. Built using the latest advances in natural language processing, OE High Impact predicts an article’s future number of citations better than impact factor alone.
Continue



 LOGIN
LOGIN

Join the Conversation
Please Login or Join to leave comments.