Efficacy of a cannabis-based oromucosal spray in managing peripheral neuropathic pain

Efficacy of a cannabis-based oromucosal spray in managing peripheral neuropathic pain
A double-blind, randomized, placebo-controlled, parallel group study of THC/CBD spray in peripheral neuropathic pain treatment
Eur J Pain. 2014 Aug;18(7):999-1012. doi: 10.1002/j.1532-2149.2013.00445.xSynopsis
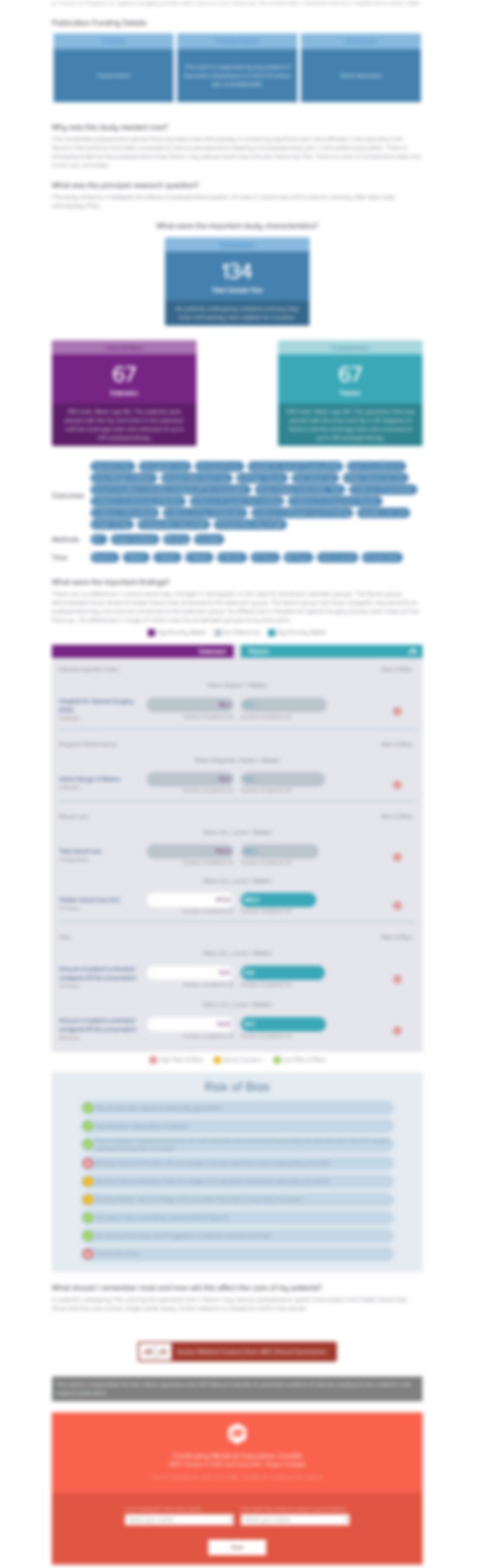
303 patients with peripheral neuropathic pain (PNP) were randomized to either Nabiximols or placebo for 14 weeks. Significant improvements in pain, quality of sleep and Subject Global Impression of Change (SGIC) were found in patients that received Nabiximols compared to those that received placebo. No patients experienced severe treatment-related adverse events in the Sativex group versus one in ...
To view the full content, login to your account,
or start your 30-day FREE Trial today.
FREE TRIAL
LOGIN
Forgot Password?
Explore some of our unlocked ACE Reports below!
Learn about our AI Driven
High Impact Search Feature
Our AI driven High Impact metric calculates the impact an article will have by considering both the publishing journal and the content of the article itself. Built using the latest advances in natural language processing, OE High Impact predicts an article’s future number of citations better than impact factor alone.
Continue



 LOGIN
LOGIN

Join the Conversation
Please Login or Join to leave comments.