
WCO-IOF 2016: Daily subcutaneous abaloparatide vs teriparatide & placebo for osteoporosis

WCO-IOF 2016: Daily subcutaneous abaloparatide vs teriparatide & placebo for osteoporosis
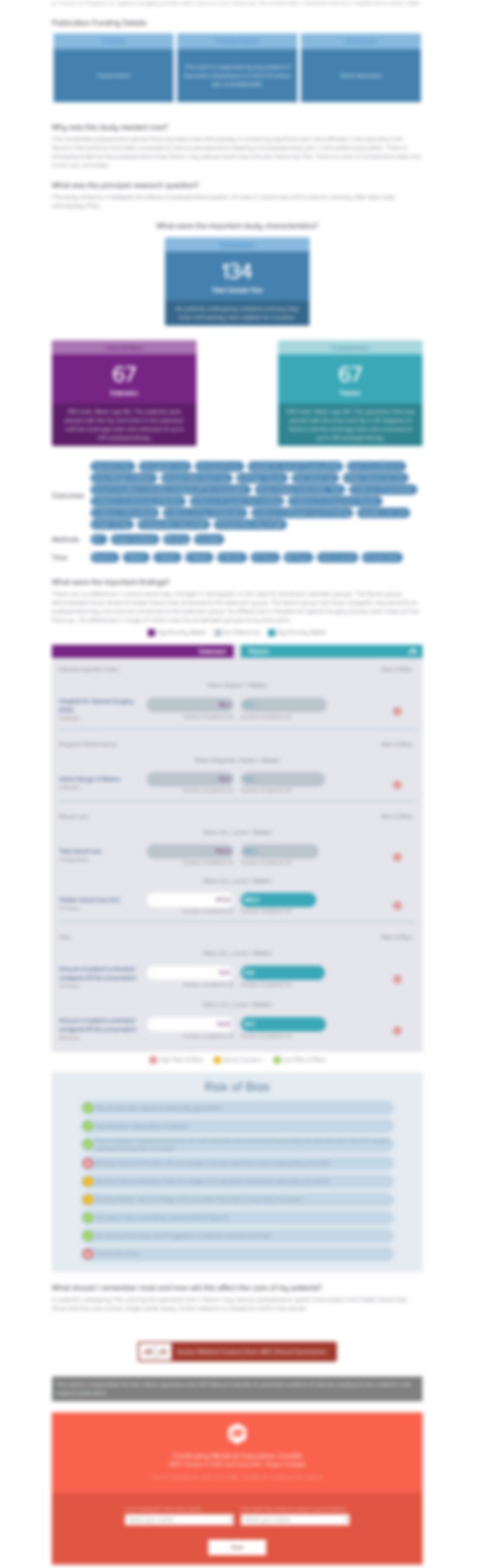
Effects of abaloparatide on vertebral, nonvertebral, major osteoporotic and clinical fracture incidence in postmenopausal women with osteoporosis: result of the phase 3 ACTIVE trial
CONFERENCE ACE REPORTS
This ACE Report is a summary of a conference presentation or abstract. The information provided has limited the ability to provide an accurate assessment of the risk of bias or the overall quality. Please interpret the results with caution as trials may be in progress and select results may have been presented.
Synopsis
2463 postmenopausal women with osteoporosis were randomized to one of three groups: daily subcutaneous abaloparatide (80ug), daily subcutaneous teriparatide (20ug), or daily subcutaneous placebo. The treatment period was 18 months, and patients were assessed for bone mineral density and incident fracture rate. Abaloparatide treatment was reported to result in a significantly greater increase in bo...
To view the full content, login to your account,
or start your 30-day FREE Trial today.
FREE TRIAL
LOGIN
Forgot Password?
Explore some of our unlocked ACE Reports below!
Learn about our AI Driven
High Impact Search Feature
Our AI driven High Impact metric calculates the impact an article will have by considering both the publishing journal and the content of the article itself. Built using the latest advances in natural language processing, OE High Impact predicts an article’s future number of citations better than impact factor alone.
Continue



 LOGIN
LOGIN

Join the Conversation
Please Login or Join to leave comments.