Zoledronic acid reduces number of skeletal-related events in new multiple myeloma patients

Zoledronic acid reduces number of skeletal-related events in new multiple myeloma patients
Effects of zoledronic acid versus clodronic acid on skeletal morbidity in patients with newly diagnosed multiple myeloma (MRC Myeloma IX): secondary outcomes from a randomised controlled trial
Lancet Oncol. 2011 Aug;12(8):743-52. doi: 10.1016/S1470-2045(11)70157-7. Epub 2011 Jul 21Did you know you're eligible to earn 0.5 CME credits for reading this report? Click Here
Synopsis
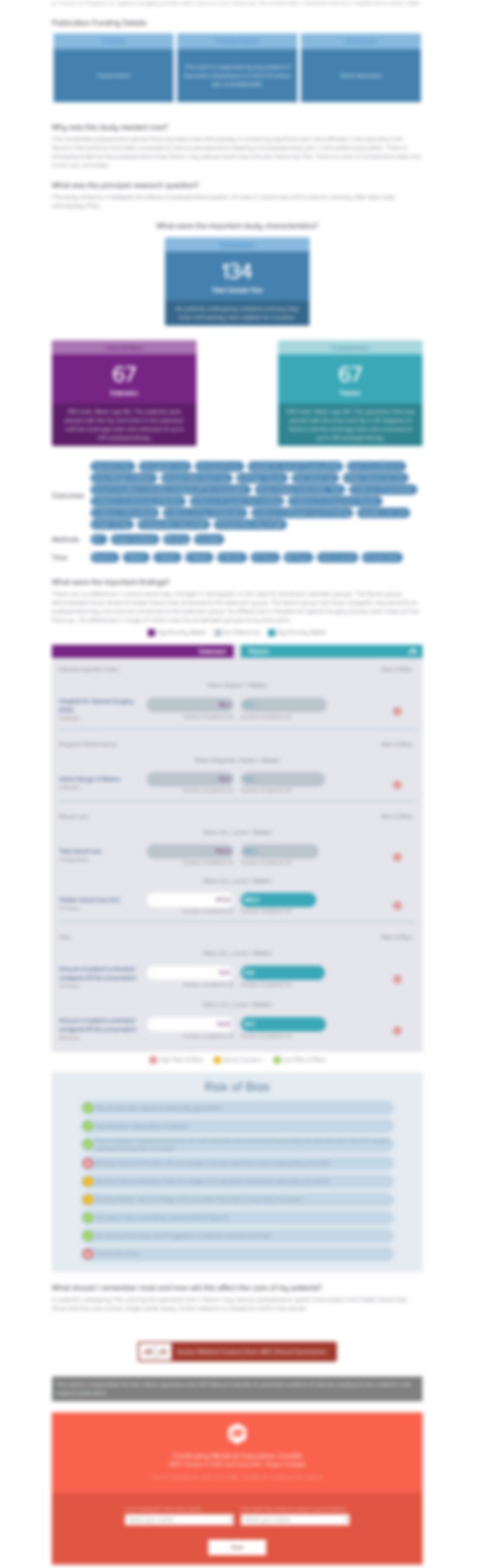
1960 patients with multiple myeloma were randomized to undergo either intensive or non-intensive treatment. Within each treatment type patients were further randomized to receive either zoledronic acid intravenously or clodronic acid orally, in order to determine which bisphosphonate and treatment type was more effective in lowering the danger of skeletal-related events. Following assessments over...
To view the full content, login to your account,
or start your 30-day FREE Trial today.
FREE TRIAL
LOGIN
Forgot Password?
Explore some of our unlocked ACE Reports below!
Learn about our AI Driven
High Impact Search Feature
Our AI driven High Impact metric calculates the impact an article will have by considering both the publishing journal and the content of the article itself. Built using the latest advances in natural language processing, OE High Impact predicts an article’s future number of citations better than impact factor alone.
Continue



 LOGIN
LOGIN

Join the Conversation
Please Login or Join to leave comments.