
Zoledronic Acid for Nonmalignant Painful Bone Marrow Lesions

Zoledronic Acid for Nonmalignant Painful Bone Marrow Lesions
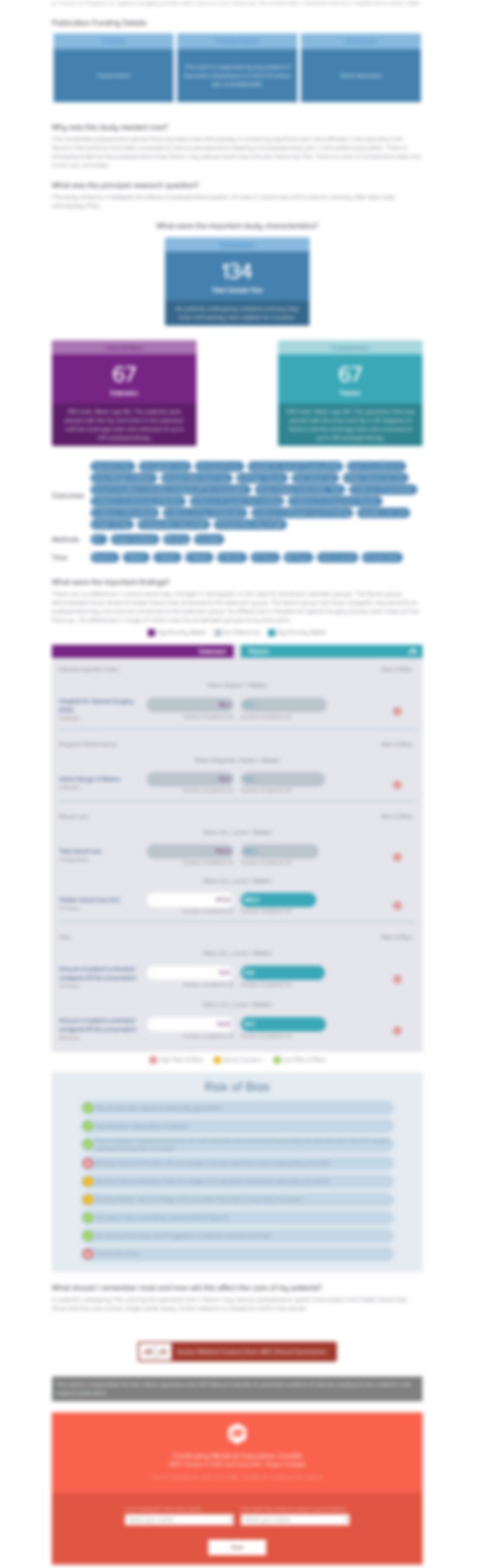
Efficacy of Zoledronic Acid in the Treatment of Nonmalignant Painful Bone Marrow Lesions: A Triple-Blind, Randomized, Placebo-Controlled Phase III Clinical Trial (ZoMARS)
J Bone Miner Res. 2022 Mar;37(3): 420-427.Synopsis
48 patients with painful bone marrow lesions were randomized to receive Vitamin D and calcium supplementation plus a single dose of intravenous Zoledronic acid (n=34) or placebo (n=14). The primary outcome of interest was the change in volume/size of the lesion. Secondary outcomes of interest included pain on a Visual Analog Scale (VAS), pain disability index (PDI), bone-specific quality of life q...
To view the full content, login to your account,
or start your 30-day FREE Trial today.
FREE TRIAL
LOGIN
Forgot Password?
Explore some of our unlocked ACE Reports below!
Learn about our AI Driven
High Impact Search Feature
Our AI driven High Impact metric calculates the impact an article will have by considering both the publishing journal and the content of the article itself. Built using the latest advances in natural language processing, OE High Impact predicts an article’s future number of citations better than impact factor alone.
Continue



 LOGIN
LOGIN

Join the Conversation
Please Login or Join to leave comments.