
OTA 2019: Feasibility of including cognitively impaired patients in an RCT on hip fractures

Did you know you're eligible to earn 0.5 CME credits for reading this report? Click Here
CONFERENCE ACE REPORTS
This ACE Report is a summary of a conference presentation or abstract. The information provided has limited the ability to provide an accurate assessment of the risk of bias or the overall quality. Please interpret the results with caution as trials may be in progress and select results may have been presented.
Synopsis
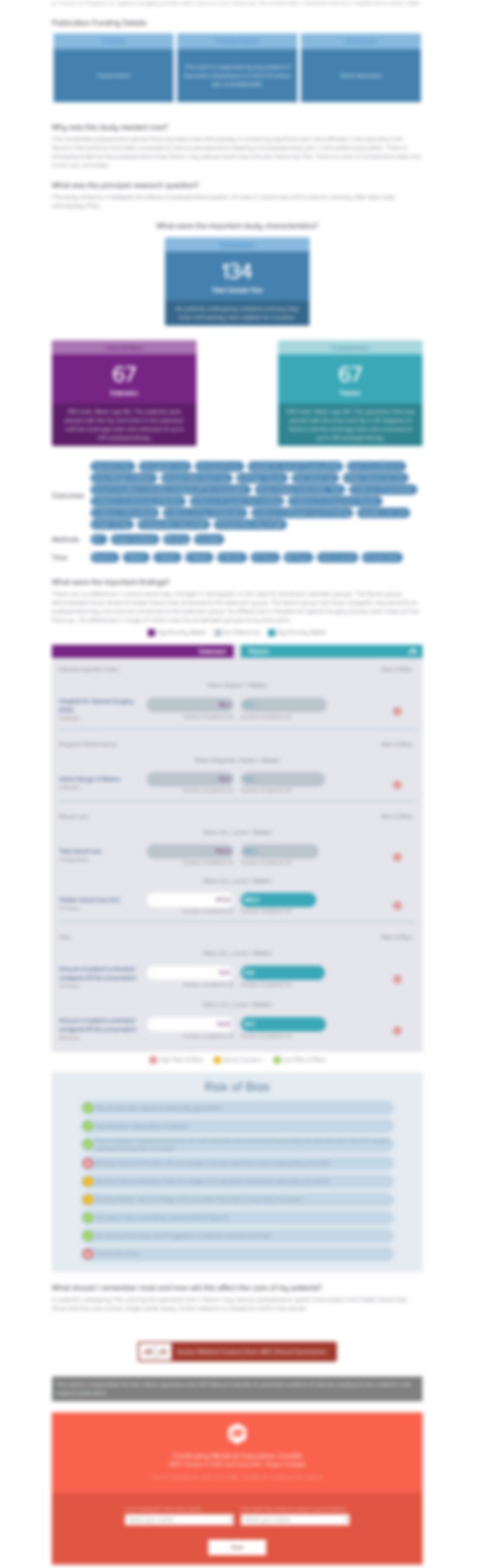
60 patients between the ages of 55-95 with low-energy intertrochanteric unstable proximal femoral fractures were randomized to receive a dynamic hip screw or intramedullary nail. The primary outcome of interest was the performance of the timed up and go (TUG) test, measured at 2, 4 and 12 weeks. Patients completed the abbreviated mental test score (AMTS) to determine the level of cognitive impairm...
To view the full content, login to your account,
or start your 30-day FREE Trial today.
FREE TRIAL
LOGIN
Forgot Password?
Explore some of our unlocked ACE Reports below!
Learn about our AI Driven
High Impact Search Feature
Our AI driven High Impact metric calculates the impact an article will have by considering both the publishing journal and the content of the article itself. Built using the latest advances in natural language processing, OE High Impact predicts an article’s future number of citations better than impact factor alone.
Continue



 LOGIN
LOGIN

Join the Conversation
Please Login or Join to leave comments.