
Topical TXA superior to placebo in reducing blood loss after instrumented posterior spinal fusion

Topical TXA superior to placebo in reducing blood loss after instrumented posterior spinal fusion
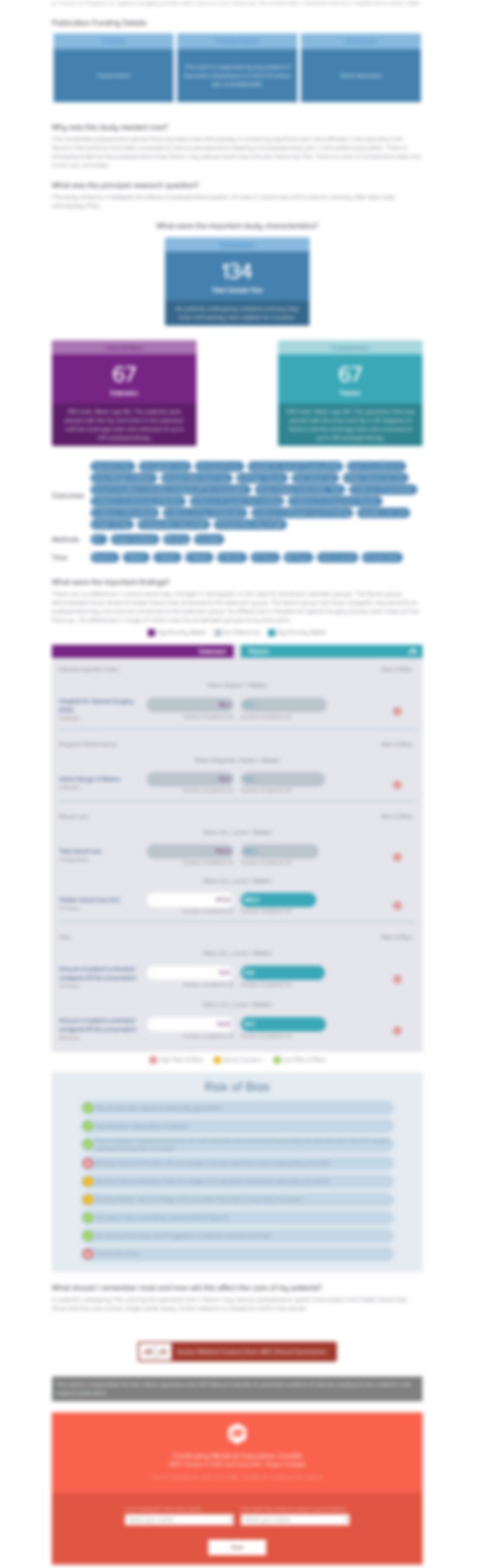
A Randomized Controlled Trial of Topical Application of Tranexamic Acid in Patients with Thoracolumbar Spine Trauma Undergoing Long-Segment Instrumented Posterior Spinal Fusion
Asian Spine J. 2019 Feb;13(1):146-154.Did you know you're eligible to earn 0.5 CME credits for reading this report? Click Here
Synopsis
60 patients between the ages of 18-70 with thoracolumbar spinal injuries and scheduled for long segment instrumented posterior spinal fusion surgeries with local autologous bone grafts were randomized to receive topical tranexamic acid (TXA) or a placebo solution for the control of blood loss and need for transfusion. The primary outcome of interest was post-operative packed red blood cell transfu...
To view the full content, login to your account,
or start your 30-day FREE Trial today.
FREE TRIAL
LOGIN
Forgot Password?
Explore some of our unlocked ACE Reports below!
Learn about our AI Driven
High Impact Search Feature
Our AI driven High Impact metric calculates the impact an article will have by considering both the publishing journal and the content of the article itself. Built using the latest advances in natural language processing, OE High Impact predicts an article’s future number of citations better than impact factor alone.
Continue



 LOGIN
LOGIN

Join the Conversation
Please Login or Join to leave comments.